|
 |
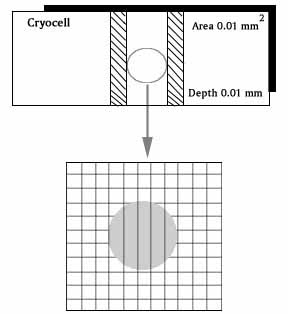
Cryocell
The Cryocell Sperm counting chamber is a simple to use device for rapid and accurate Sperm count & motility evaluation directly from undiluted Semen. 10 MICRON DEPTHDESCRIPTIONThe Chamber is composed of two parts: The lower main part has a glass base wherein the centre portion has been coated with a metal layer on which there is grid of 0.01 sq. mm. This part has been lowered by 10 microns as compared to the rest of the chamber. It is on this portion that a very small drop of the sample is placed. Figure:
The upper part is cover glass, which is to placed on the drop of the sample & firmly pressed on both the sides to remove the excessive sample, thereby ensuring that the space between the cover slip & the Grid is exactly 10 microns. PREPRATION OF THE CHAMBER Before use, make certain that the opposed glass surfaces are absolutely clean and free of dust, since the size of the particles is more than 10 microns. You should brush the surfaces with a very soft brush.(Provided with the pack) This is a high precision instrument built to very narrow tolerances. Thus extra care & cleanliness is advised. The depth of the chamber is guaranteed at 10.0 microns ( + 0.001 microns). Any speck of dust deposited on each side of the chamber or in chamber itself can render this accuracy void. Please therefore carefully clean all surfaces of the chamber & the cover slip with water and a brush & thereafter with special Lens paper & a dry brush. SEMEN ANAYSIS Mix the specimen well, taking care to avoid formation of bubbles, with the aid of a rod, put a small drop in the centre of the centre strip. Grasp the cover glass & Place it on the edges & press firmly. This ensures the automatic spread of the drop into the thickness of 10 microns. It is recommended that the drop should spread on the entire area of the centre portion. Once the cover glass is in place avoid further touching. Lifting and recovering as this may affect the uniform spread of the sperm within the drop. Lift the chamber & place is on the stage of the microscope using a 20x objective and a 10x eyepiece. Avoid using the 10x objective, as the sperms with e too small to see. After focusing move the stage of the microscope and locate the grid in the centre of the view area. Uneven distribution of the Sperms mean that the sample was not mixed thoroughly. If sperms cannot be seen in one focal plane, improper cleaning of the glass surfaces is suspected. In either case- repeat this procedure after correcting he fault. MOTILITY ESTIMATION Count all the non- motile sperms within (10 continuous squares either horizontally or vertically). Then count the motile sperms in the same area. Adding up both the numbers would give you the total count per ml.( in Millions). You can then divide the motile with the total count to reach the percentage motility. Using the same method estimate the grade of the motility from +1 to +4. SPECIAL CASES Bubbles : If bubbles are present in the grid area, it is recommended that the drop may be replaced by another, unless the bubbles are small enough not to interfere with the analysis. Large particles of dust, threads etc. will also interfere with the count by changing the depth of the space & in such a case the drop should be replaced. Major variation in counts between drops of the same specimen means, that the sample was not mixes well or is too viscous to mix completely. CLEANING AND PREPRATION FOR REUSE Dip the brush in to clean water and simply wipe both sides of the glasses. Then squeeze the brush and sponge the remaining water off the chamber. Finally dry the surfaces with special lens paper.(Provided with the pack) The Chamber is now ready for re- use. |